WE ARE THE SMARTICLE PARTICLES. We are composed of three pretty awesome people named Melissa Jessica and Hanae.
Tuesday, December 14, 2010
The Magnificent LAB 4C :)
Before I tell you about the lab, ther first thing you need to know is what a hydrate is. A hydrate is a compound that has a definite number of water molecules incorporated into its crystal structure. The crystals appear dry, but when these compouds are heated strongly, water is given off, leaving the anyhydrous form of the compound.
The first mass we found was the empty crucible. We weighed the crucible on a centigram scale. The mass my partner and I found was 25.23g. Then we found the mass of the crucible and the hydrate, which was 30.11g.
To find the mass of the hydrate alone, this is the formula:
Mass of crucible and hydrate - Mass of empty crucible = Mass of hydrate
30.11g - 25.23g = 4.88g
Next, we heated the crucible and anhydrous salt using a bunsen burner and weighed it again on the centigram scale. The mass we found was 27.79g. After that was found, we heated the same objects again for a second heating and came up with a mass of 27.78g
To find the mass of the anhydrous salt alone, this is the formula:
Mass of the first heating - Mass of the empty crucible = Mass of anhydrous salt
Lastly, to find the mass of the water given off, this is the formula:
Mass of the hydrate - Mass of the anhydrous salt = Mass of the water given off
ANALYSIS OF RESULTS (whopee)
1. Calculate the percentage of water in a hydrate
%H20 = 2.33g
---------- x 100% = 47.7%
4.88g
2. Calculate the number of moles of the anhydrous salt left behind.
2.55g x 1mole
--------------- = 0.02 mol
120.4g/mol
3. Calculate the number moles of water removed by heat from your sample of hydrate.
2.33g x 1 mol
----------- = 0.13 mol
18g/mol
4. Calculate the moles of water per mole of the anhydrous salt.
0.02
------ -> 1
0.02
0.13
------ -> 7
0.02
5. What is the empirical dormula of the hydrate?
AB . 7H2O
Thursday, December 2, 2010
Empirical & Molecular Formula
Empirical Formula: Gives the lowest whole number ratio of atoms in a the formula.
Molecular Formula: A multiple of the empirical formula that shows the actual number of atoms that combine to form a molecule. (I will show you how to determine what the multiple is later on.)
Let's being with the empirical formula...
What is the empirical formula of a compound consisting of 60.0% C and 40.0% H?
We are going to assume that we have 100g of the compound, because 80% + 20% works out to 100%. It is the ratio that counts, so it's okay if we don't really have 100g- the ratio will still be the same as long as the percentages are the same.
1) Find the number of moles of each element...

Wednesday, December 1, 2010
How to Calculate Percentage Composition (The Fun Way)
Hey It's Jessica Here from the Smarticle Particles! I hope you like this video I made. This is how you calculate Percentage Composition in a non-boring way : ) enjoy.
Tuesday, November 23, 2010
Mole Conversions for the Gifted (Self-Made Video Surprise Included!)
Harriet, played by Hanae Aso.
Savannah, played by Melissa Chan.
JiaLiu, played by Jessica Tsui.
Please enjoy this fun and entertaining video!
Sunday, November 21, 2010
The All Powerful MOLE
Today we will be focusing on this little strange creature right here:

Don't you think this creature is fascinating????
Why yes is, you reply, but I don't think this is what we're supposed to be focusing on. AND YOU ARE COMPLETELY RIGHT.
Actually, what we learned in class was mole conversions. In order to completely totally utterly understand conversions, you must know the mole map which I will oh so kindly show you:

Converting PARTICLES <-> MOLES
Remember, there are 6.022x10²³ particles in one mole
6.022x10²³particles
1 mole
So lets have an example now shall we?
Say there are 6 x 10^24 particles of carbon and somebody out there really wants you to convert that into moles. What will you do? I'll tell you:
6 x 10^24 particles x 1 mole/ 6.022 x 10²³ = 9.96 moles of carbon
Converting MOLES <-> PARTICLES/MOLECULES/FORMULA UNITS/ATOMS
Lets have an example:
Say that you have 4 moles of water (H20)and you NEEDED to find how many molecules there were within the water you had. Here is how you do it:
4 moles x 6.022 x 10²³ molecules/ 1 mole = 2.4 x 10^24 molecules of H20
LAST BUT NOT LEAST. Hold on to your horses guys, we're almost done.
Converting MOLES <-> GRAMS
Last example of the blog:
For example, you are given 1.5 moles of carbon and you needed to change that into grams. This is what you must do to gain ultimate world domination:
1.5 moles x 12.0 g/ 1 mole = 18.0g of Carbon
And there you have it folks.

Friday, November 12, 2010
Lorenzo Romano Amedeo Carlo Bernadette Avogadro di Quaregna e Cerreto
Lorenzo Romano Amedeo Carlo Bernadette Avogadro di Quaregna e Cerreto
No jokes!
Hello everyone, this is Melissa from the Smarticle Particles. I hope you enjoyed the above photo of that dashing and handsome man!
Just for the ease of typing, we are going to call him Avogadro for the duration of this entry. Anyways, this was his hypothesis...
- Equal volumes of different gases will have the same number of particles as long as they have the same temperature, and pressure.
- If they have the same number of particles, then the mass ratio is due to the mass of particles.
(Use this hypothesis for the relative masses of all atoms on the periodic table!)
Let us introduce you to the following 4 types of masses. You are going to have to know these by heart.
ATOMIC MASS:
The mass of 1 atom of an element in Atomic Mass Units (AMUs).
Ex/ Fluorine = 19.0 AMUs
FORMULA MASS:
All atoms of a formula of any ionic compound. So basically the masses of all the elements in a compound added together.
Ex/ Potassium fluoride.
K F
39.1 + 19.0 = 58.1 amu
MOLECULAR MASS:
Basically the same as formula mass EXCEPT it is regarding covalent compounds. YA DIG? So just simply add all of the masses of the elements in the compound. I know it sounds confuzzling, but it's really not that bad after The Smarticle Particles explain it to you.
Ex/ Carbon monoxide
C O
12.0 + 16.0 = 28.0amu
MOLAR MASS:
So um yeah, chemistry just gave us another confusing term that really isn't that complicated. It's essentially ANY atomic/molecular/formula mass, added and garnished with a g/mol.
Ex/ 1 mole chlorine = 35.5g/mol
1 mole fluorine = 19.0g/mol
....YA DIG?
AVOGADRO'S NUMBAAAAA
Okay. This number is really big. Like. You won't even know it until you see it. We. Mean. BIG.
6.022x10²³
This number also happens to be the number of particles in 1 mole of any substance.
6.022x10²³ particles/mol
This number allows for atoms and molecules to be counted!
Tuesday, November 9, 2010
Take your entrance exam for "Canada's Next Top Chemist"!
You are going to want to be comfortable with:
- Significant Figures
- Measurement + Uncertainty
- Scientific Notation
- Density
- Graphing
- Lab 2E
- Unit Conversions
- Formulas for Volume, Area, Density, and Percent Experimental Error.

Sunday, November 7, 2010
Even Microsoft Excel Needs Some Love!
"I just feel so lonely- I know I can be confusing and hard to understand, but once you get to know me I'm really not that bad. *cries*" - Microsoft Excel
Do you see how negatively our behavior has affected Excel? That is why today we are going to learn more about Excel so it doesn't feel so lonely anymore. To be more specific, we are going to learn how to graph collected data, generate its equation, and determine the slope of the graph.
Just watch this quick video:
We suggest that you play around with a little and try to bling out your graph to make it more attrctive.
Friday, November 5, 2010
LAB DAY: Aluminum Foil and All That Jazz
What many people don’t realize is that aluminum is practically 100 percent recyclable. It is extremely durable and can be reused over and over again. Aluminum foil is technically just as recyclable as aluminum cans. The problem is that aluminum foil is often dirtier, thus making it harder to recycle.

ALRIGHT, now we're moving on to something thats relevant. OOOO. So basically what this blog entails is the summary of what we did in.....dun dun dun......LAB 2E.
Okay just kidding guys, it's not actually scary, so come out from under your blankets and lets get rolling.
The basic formulas you need to know are:
Density = Mass/Volume
Volume = Mass/Density
Mass = Density x Volume
Volume = Length x Width x Height (a.k.a thickness)
Height = Volume/(Length x Width)
Now I bet you're going WOAH THERE slow down my brain is imploding. Well I'm sorry, but we must move on.
BASICALLY, what we did in the lab was take 3 pieces of rectangular square-ish paper and measured each's length and width to the right amount of sig figs. (Refer back to our sig fig lesson if you are a confused child right now). We recorded them all on the chart with their absolute uncertainty.
NEXT we weighed the pieces of aluminum on a centigram scale.

Then guess what we did? We recorded it on our table!!!! Now, this is a mandatory step for if you don't record, then all your work will be lost which usually leads to tears. But dry your tears, because it is hard to read small print when you are crying and I must finish todays lesson.
After this, we had to find the exponential error. What is this you ask? Well lucky for you, I will tell you. Exponential error is the exponential growth of an error, or how an error can compound itself over time. The formula for this foreign topic is:
Exponential error = your measurement - accepted value / accepted value x 100%
NOTE: EXPONENTIAL ERROR MUST ALWAYS BE WRITTEN AS A PERCENT. If not, barney will come after you and continuously sing children songs until you become insane.............okay maybe not the worst that could happen would be you would lose marks. But nowadays, marks are EVERYTHING.
Lastly, we had to do some follow up questions. THIS is where the equations come in. Just make sure that when they ask you to find the thickness of the aluminum, it is actually the height, so use the height formula. YES, they trick you like that and YES they probably find it funny that students are pulling their hair out to try to figure out the thickness.
So I guess this is all for now folks. Hugs and hearts.

VISIT THIS LINK FOR A SPECIAL SURPRISE!
Tuesday, November 2, 2010
Density! Something Easy And Short. WIN
Ex/ What is the density of 40g of copper, with the volume of 10cm³?
D=m/v=40g/10cm=4g/cm³. I understand this is confusing, (Blogger gives me limited features. Yes, excuses, I know, I'm full of them) but all you really have to do is divide a given mass by a given volume.
Ex2/ BUT BUT BUT BUT BUT. WHAT IF WE DON'T HAVE THE MASS OR VOLUME?!!!!! Oh gosh, I'm starting to hyperventilate just by the thought of it. I'm getting frantic too. BUT don't fear! All you have to do is switch things around here and there. What if you wanted to find the volume given the density and mass?
Since the general formula for density is d=m/v , using simple algebra, in order to find volume, we know v=m/d. Therefore, now all you have to do is plug the numbers in. I'm sure you all have learnt this skill in Math 10.
When something is more dense than another object, that object will sink. If something is less dense than another object, the less denser object will float on top of another. Oil and water is an excellent example. The oil will always float on top of the water no matter what, since oil is less dense than water. Here is a cool picture. YAY!

Monday, November 1, 2010
Meh...... So Which One Was Accurate?

Let's just settle with 1 armspan=1m. You settle with the length as 26 armspans because... Well, you counted 26 armspans. Now let's say we now travel to the future and use some sort of metre-sticko-whiz. And the number comes out as 29.283928347817341203497m. So which is accurate and which is precise? Here is your answer:
When something is accurate, the value/number is CLOSE to the actual measurement. So essentially it is the correctness of a measurement. However if something is precise, it is a reproducible measurement, and usually has more significant digits.

So what's the answer? 26 armspans is accurate, and 29.283928.......m is precise. An example of an accurate YET precise reading would be 28.8671023934871m.
NEW TOPIC!
Like how everything in this world isn't perfect, no measurement is perfect. Which concludes that no measurement is exact. It is only when we count (tangible) objects, we get exact measurements. For example, 30 people in the class. We cannot have 29.5 people, can we? That would just be awkward.
Absolute uncertainty is the expressed uncertainty in a certain measurement. So how on earth do we do something like that? Luckily, there are two methods.
Method number glorious one. Make at least three measurements when measuring whatever. Then calculate the average of all your measurements; The absolute uncertainty is the largest range between the lowest measurement measured and the highest. However there is one catch--You MUST throw away, crumble up, burn to ashes, sautee, and sprinkle over Donald Trump's combover, numbers that do not "belong". Let's say you have four measurements. 1.) 16.88 2.)19.89 3.)16.50 4.)16.94. Which is the odd one out? Obviously measurement number two must be discarded.
Method number two. Just simply determine the uncertainty of each measurement you make. Just use the eyes that God gave you, and use them to the best of your ability, and measure as precisely and accurately as you can. You should estimate at least to the 10th of a fraction of each increment on a measuring utensil. For example if a test tube went up in increments of 1, your uncertainty would be +- 0.1.
RELATIVE UNCERTAINTY This HAS to be expressed as a percent. (I learnt it the hard way by getting a mark off my assignment). The number of sig figs in a measurement indicates the relative uncertainty. Remember that the number of significant figures all certain digits plus all uncertain digits. There is a general formula for relative uncertainty: (Absolute uncertainty/estimated measurement) x 100
Wednesday, October 27, 2010
Seperating All The Wonderful Glories of Chemistry
So today we will be learning about the joys of Separation Techniques. There are five main types we will be learning about today:
-Filtration
-Floatation
-Crystallization and Extraction
-Distillation
-Chromatography.
Scratch that, there are six, I think.
Let's first talk about Hand Separation and Evaporation. A few key points to remember:
-This is when you try and separate solids from solids.
-Magnetic things can be separated using a magnet.
Ex; There are a bunch of coins and you want to separate the pennies from everything else.
- You'd obviously just use your hands and pick the pennies out.
-Evaporation, I'm sure you all know is when you heat up liquids so you get the solid residue/remains.
-Evaporation is used when there is a solid dissolved in liquid solution.
Ex; When you have salt water and you only want the salt.
Second method, (gosh I'm tired already) Filtration
-You can use filtration when there is an undissolved solid inside a liquid.
-You'd use a porous filter to separate the two matters.
-If the pores of the filter are smaller than the particles themselves, obviously the solid stuff will stay on the filter while all the gas and liquid passes through it; solid particles are too rigid to pass through the pores.
-You'd usually use filter paper to do all this.
Third method (trust me, this kills me as much as it kills you), Crystallization
-Use this method when you are separating a solid from a liquid.
-First you'd have to convert the solid into a solute. The solute is usually made via a chemical or physical change.
-After converting the solid into a solute, you'd then separate it using the filtration or floatation method.
-Finally, you evaporate or cool down the substance until the solid comes out as pure crystals. If needed, you can filter the crystals from the remaining solvent.
Fourth Method (I should really get to PHYSICS soon...), Gravity Separation
-This is a pretty straightforward method.
-When you use gravity to separate things based on their density.
-Usually used to separate solids from solids.
-Something called a centrifuge helps do this, and whirls a test tube around at a super high speed making the denser substance sink to the bottom.
-Works most effectively when done with small amounts of material.
Fifth Method! (back to enthusiasm! Yay chem!), Solvent Extraction.
-This is when a component wafts into a solvent when it is shaken with the mixture.
-This works effectively when done with solvents that only dissolve one component.
-When it is a MechanicalMixture, a solid mixed with a solid, you use liquid to dissolve ONE of the solids. The solid is then left behind or dissolved.
-But oh, what if the solvent is insoluble?
-The liquid will then form layers and drain the insoluble solvent.
-This leaves the unwanted substance (solid) behind.
Sixth Method (Not going to lie,I just got distracted by Facebook), Distillation.
-Have you ever seen "distilled water" on your water bottle? Well I'm going to educate you about that.
-You heat the mixture, and substances with a low boiling point vapourizes first.
-Essentially, it is the collecting and condensing of vaporized components.
<---------Diagram.
Seventh and final Method!!...Split into two parts. Chromatography.
-Two types of chromatography:
-Paper Chromatography (PC)
-Thin Layer Chromatography (TLC)
-A mobile phase wooshes the sample over a stationary phase. (graphically speaking)
-Separates complex mixtures like drugs, plastics.
-A highly accurate and precise method.
-Once separated, you can collect the components individually. So basically, it's convenient.
Sheet Chromatography.
-PAPER CHROMATOGRAPHY:
-Stationary Phase is a liquid drenched into a sheet of paper.
-Mobile Phase is simply a liquid solvent.
-Some components take longer in the stationary phase.
-Appears in separate spots on the paper after separation is complete. Or after "developing"
-THIN LAYER CHROMATOGRAPHY:
-Stationary Phase; a very thin layer of absorbent. It usually coats some sort of sheet.
-Ditto Paper Chromatography, the "developed products" appear on various spots on the sheet after procedure is complete.
Monday, October 25, 2010
Read on for the Scariest and most FREAKY story you have ever heard. It is called....THE NAMING OF ACIDS.
Well my friends..............I am having that exact problem right now. But, I will try. Feel free to add in any shrieks and hollers for EMPHASIS...a.k.a making me feel better. ALL RIGHT let's get started
*puts flashlight to face*
It was a dark grey spookey night. _______ (insert your name here) was wandering through the woods ALONE at night on halloween. (You aren't that smart I guess)

Every step you take leads you closer to the dangers that lie ahead. You ignore the noises you hear from the bushes beside you..thinking it is just the wind, BUT soon enough you realize, THERE IS NO WIND.
You whip around looking everywhere to see who is the source of the noise, then suddenly, a man POPS OUT OF THE BUSHES.
He has the word "Acid" printed across his shirt.
"Who are you?" you say trembling.
"Why, I am ACID MAN!" the guys screams. "I CORRODE AND BURN YOUR SKIN AND TASTE SOUR"
"NOOOOO" you scream. "Is there any way I can convince you not to burn me?"
"Well, there is ONE way, but NO ONE has completed this task before. All you have to do, is explain to me the different ways of naming SIMPLE and COMPLEX acids. Then I will let you free. You see, no one knows how to name me nowadays." he says.
"I will take that challenge" You reply, remembering that you have learned this lesson already in chemistry class.
So, those who do not know how to name acids will get burned. For those of you that do, GOOD JOB, you are free to go : D
Okay fine, I'll help you guys out, after all, we need people to continue reading our blog, we can't just let acid man burn all of you. pshht.
An acid is a covalent bond that is formed by a hydrogen ion and a negatively charged ion dissolved in water. The ions separate when they are dissolved in water.
Chemical formulas for acids, start with the letter H which stands for? No its not habanera, harmonometer, or hygeiolatry (if you guys even know what those mean..I do..hehe). Well the letter H stands for Hydrogen in the formula.

First, let's start by naming simple acids.
These always start using the prefix: hydro
But after hydro, you need the name of the negatively charged ion. You can't have something like hydrobiological and say its an acid.
Examples of simple acids are:
HBr
HS
HF
Now, to name these, the negatively charged ions need to end using "ic" then write acid after this. Soo...
HBr = Hydrobromic acid
HS = Hydrosulphuric acid
HF = Hydrofluoric acid
Simple enough?
Now we're moving on to the complex acids. This is where it starts getting tricky.
If the polyatomic anion ends in "ate" you need to change the end to "ic"
If the polyatomic anion ends in "ite" you need to change the end to "ous"
An EASY was to remember this is to say in your head:
I "ate" "ic" and got "ite" "ous"
orr what we learned in ms chen's class
I "ate" "ic"-y sushi and got appendic "ite" "ous"
Oh wait, they're basically the same...but one's longer. Oh well.
Some examples of complex acids are:
HNO3
HNO2
HCH3COO
Now to name these, instead of starting with hydro, you need to start with the name of the polyatomic anion with the new ending then write acid after it. SO:
HNO3 = Nitric Acid
HNO2 = Nitrous Acid
HCH3COO = Acetic Acid
The only exception to these rules is the acid HCN. This is a simple acid so it is named hydrocyanic acid.
NOW, TELL THAT TO THE ACID MAN.
Well, my fingers are tired now, so basically you know what happens next in the story. You tell acid man and he lets you free then you live happily ever after yada yada yada.
THE END.
Wednesday, October 20, 2010
A Fun, Fantastic, and Fulfilling Day with PAPER CHROMATOGRAPHY!
Ooooh. Sorry. Did I cross the line? Okay, I'll stop. :S
THE GENUINE AND SOPHISTICATED LAB SUMMARY BEGINS NOW.
On Tuesday, October 19th, 2010. We performed Lab 3B out of Essential Experiments for Chemistry. This is page 32-37 of the book.
The idea of the lab was to discover the components and Front Ratio (Rf) of various solutes (food coloring) with a solvent (water). We were to do this through paper chromatography, of course.
I hope that you all remember what was done during the lab. For example, the observations you made, the type of data collected, and the calculations made.
Recall that we drew a pencil line across each of our three 22cm strips of chromatography paper, 4 cm from the end, and cut it so that the strip has a pointed tip.
After putting a drop of sample onto the drawn line of the strip, we placed it into a test tube with 2cm of water. After 20 minutes of waiting, we measured the distance between the length travelled by the solute (d1) by the solvent (d2) from the pencil line. (Measurements were done using a ruler, measuring in cm) We then calculated the component:solvent front ratio (Rf).
The way to calculate the front ratio was simply to divide the distance that the solute travelled by the distance that the solvent traveled. Your result (aka. your Front Ratio, Rf) should be between 0 to 1. This "rule" can be found in your lab book!
Remember all the observations you saw while the fronts moved. Certain colors of the solute seperated into multiple colors, such as blue, yellow, and red! However, some solutes, such as the one with yellow coloring- did not seperate! It just remained the same color except expanded. This is because there are no colors that mix together to create yellow. But, yellow can be found as a component, because it is used in mixing to create new colors.
This lab should've helped you understand a very accurate form of seperation, which again, is called paper chromatography!

Monday, October 18, 2010
Review from Science 10 when we were Oh So young and uncivilized: Writing + Naming Ionic and Covalent Compounds
The lesson today was on writing and naming Ionic and Covalent Compounds (as you can probably plainly see from the title). Does it sound fascinating? Well GOLLY GEE it sure was.
Well, lets start off with explaining what ions are. Ions are atoms or groups of atoms that are positively or negatively charged. Basically, they are composed of two or more oppositely charged particles (which are ions). They are held together by electrostatic forces (ooo :D)
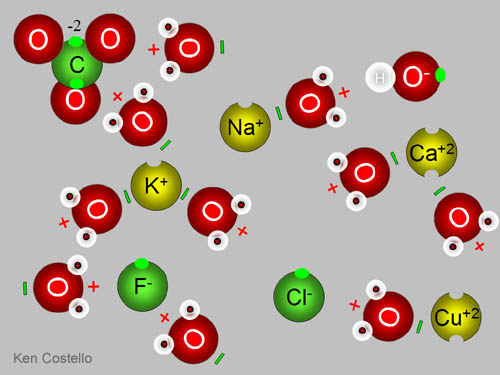
In ionic compounds, electrons are transferred from a metal to a non metal. When writing Ionic Compounds, I LOVE doing the switch method. It's the easiest to understand. Hehe. What happens is the two particles switch charges.
For Example:
Strontium Fluoride
Strontium has a charge of +2
Fluoride has a charge of -1
So, that's Sr2F. BUT since they switch charges, it becomes SrF2
Simple enough? Well too bad, now it gets harder. If the charges are the same, they both canacel out.
For example:
Calcium Sulphide
Calcium has a charge of +2
Sulphur has a charge of -2
When this happens, DO NOT (under any circumstances, even if your dog is dying) write Ca2S2. WRONG WRONG WRONG WRONG WRONG.
The two's cancel out and that makes CaS.
Lastly, you need to know what happens when you have numbers that can easily divide into eachother
For Example:
Rhodium (IV) Oxide
Rhodium has a charge of +4
Oxygen has a charge of -2
So the equation is Rh2O4. BUT *gasp* there is a catch, this formula can reduce!!!
2 can easily divide into 4 because 4/2 = 2 (I hope you already know that but if you're suddenly brain dead, I wrote it out for you. It's okay, I have brain farts all the time).
SOOO...the formula becomes RhO2 because the simplifying cancelled out the 2 on rhodium and made the 4 a 2 on oxygen.
You see how that works? MAGICAL!
So now, we can finally move on to naming Ionic Compounds =.=
Okay so just to review a bit from last year, the metal is ALWAYS first and the non-metal is ALWAYS second. (Don't you feel bad that it always has to come in second? D:)
When naming, you need to change the ending of the non-metals to "ide"
For example:
Sulphur becomes Sulphide
Oxygen becomes Oxide
Fluorine becomes Fluoride
And it goes on and on and on and ooonn, strangers, waiting, up and down the boulevaarrdd. (okay i got carried away).
When the particle has more than 1 charge, you need to state the charge in roman numerals after the name of the particle.
For example:
FeO = Iron (II) Oxide
PbCl4 = Lead (IV) Chloride
If you do not remember your roman numerals (which I totally understand) I will be kind enough to write them out for you :)
1 = I 3 = III 5 = V
2 = II 4 = IV 6 = VI
Okay guys, we're now moving on to covalent compounds oooo, ahhh, ohhh. yes.
So covalent compounds, unlike ionic compounds, are between a non metal and non metal. They share electrons. Writing covalent compounds have the same rules as writing ionic compounds, but naming them is a different story.
Covalent compounds use Greek prefixes to indicate the number of atoms. Ohoho what is this? It appears we are taking a trip back YET AGAIN to ancient greece.
To truly understand the naming of covalent compounds, we must understand the Greek ways of numbering.
Mono = 1 Penta = 5 Nona = 9
Di = 2 Hexa = 6 Deca = 10
Tri = 3 Hepta = 7
Tetra = 4 Octa = 8
You need to put the greek prefixes infront of the particles names.
For example:
NH3 = Nitrogen trihydride
You do not need to put a "mono" infront of nitrogen even though it only has one atom, because you don't need to for the first particle.
The last thing you folks need to remember is the Diatomic Molecules. These are:
Hydrogen, Oxygen, Fluorine, Bromine, Iodine, Nitrogen, and Chlorine.
OR better known as
HOFBrINCl
(prounounced Hoffbrinkle)
These all have two atoms:
H2, O2, F2, Br2, I2, N2, Cl2
When named, you must put a "gas" behind them.
For Example:
H2 = Hydrogen Gas
O2 = Oxygen Gas
F2 = Fluorine Gas
WELL THANKS FOR READING. I KNOW IT WAS A LOT. Now you may rest your eyes from staring at this screen. Researchers say it is bad to stare at a computer for more than two hours at a time. o.o
WELL BYE.
peace.
Monday, October 4, 2010
Thursday, September 30, 2010
[LAB DAY!] Chemical and Physical Changes. Y'all.
Hai y'all. As y'all know Chemistry ain't no joke ny' more. Now we ain't meanin' to be aggravatin' y'all, but we're gunna needa pull our legwarmers up and get summa that edgy-cation our folks are always talkin' bout'.
Now that you have all had your giggles and snorts. Let us dive right in.
Recall that we had 3 objectives in mind before we began the lab.
1) to observe changes while working in the lab
2) to understand why each reaction is chemical or physical
3) to record recognizable characteristics of chemical changes
Basically, this is what Ms. Chen WANTS us to gain from this lab. Ahem.
Which... we did. Right? (SAY YES.)
Well, we aren't going to write out all the procedures because normal people wouldn't have a long enough attention span to read it all. SOO we're going to sum it up for your benefit. You're welcome. :)
(Thank the Lord that you all are normal. Well... most of you all anyway.)
Because it's "important", we are going to list all the materials used in the lab... good for future reference!
- 4 small test tubes
- test-tube rack (We seriously wonder whether or not these were made in woodworking class by some unfortunate soul who had to make this boring thing as their final project!)
- 4 medicine droppers
- glass square
- lab arpon
- safety goggles
- 4 unknown solutions

Anywhooo, so we were given 4 mysterious highly toxic and illegal solutions.
(Because our well being is not a priority. Duh.)
Four solutions we labeled A, B, C, and D.
We mixed B+A, C+A, C+B, D+A, pigs+mushrooms, rats+lions, bubble tea+pottery ....... and we got a RAINBOW : D
Okay well maybe we didn't quite mix the last few but you get the point. (But wouldn't it be cool if we did?)
So, what we had to do was place all these different solutions together in a glass dish and see what kind of magical scientific reaction occured when they mixed.

If the mixture was bubbling or fizzy or changing colour, we could tell definately that it was a chemical change. So basically, anything strange that occured (like the fact that we have HST) you know its a chemical change.
If the mixture stayed the same and nothing happened to it (like you refusing to move from the couch when you're chilling and pigging out) then you know its a physical change.
Monday, September 27, 2010
We're Missing Monday Night Football =.=
Recall the three types of matter you learnt about in previous grades. Try and point out all three, using the given scenario.
Answer: The ball is a solid, the sweat trickling down his forehead is a liquid, and the air is obviously a gas.
Now wouldn't it be cool to actually go inside the ball? I think so. When inside, you see that the particles are so tightly packed that you can hardly move and you start to suffocate! You quickly move on to the liquid, his sweat. The particles are less tightly packed here, meaning that a liquid would take the shape of the container its in. Lastly, you pay attention to the air around you. You reach out and swipe at it. WOW. Magically, your hand goes through the air! The particles in the air are spread really far apart and are always moving. Gases also take the shape of the conatiner they're in.
NOW. I bet all of you ask this question daily because it is just so darn interesting.
WHAT IS MATTER??
Well my friends, matter is anything that has mass and takes up space. It is neither created nor destroyed, only changed from one form to another.
BUT WAIT! THERE'S MORE!
Matter is composed of Pure substances and Mixtures. Pure substances have one set of properties and one kind of particle. Mixtures are quite the opposite. They have more than one set of properties and substances and they are physically combined.
To make it even more difficult for students with low brain capacities and low attention spans, Pure Substances are composed of Elements and Compounds! Elements are the simplest form, meaning they cannot be decomposed and they are made of metal, non metal, and metalloid atoms. Compounds on the other hand are made of elements, they are chemically combined, and the smallest particles is an ionic or covalent molecule.
Well even though we're pretty sure we lost you, we're going to continue anyways...because we're cool kids.
WELL THEN. Mixtures are composed of two groups as well. Homogeneous and Heterogeneous. Homogeneous are uniform throughout and appear to have only one component. Heterogeneuous are not uniform and appear to have more than one component.
IT'S ALMOST OVER HANG IN THERE.
The last topic we are going to cover is Physical and Chemical Change.
Physical Change is when no new substance is formed and chamical composition does not change. Physical Change is reversible.
For example: Melting ice. You can un-melt ice meaning it is reversible.
Chemical Change is when no new substance is produced. Chemical Change is irreversible.
For Example: Cooking a steak. You cannot uncook a steak. You can try to, by putting it in the freezer or chopping off all the seemingly cooked bits and feeding them to your dog but NO. I am sorry to say you cannot un cook something that has been cooked. Therefore it is irreversible.
THANK YOU. We love you. We really do.
The Day After Visiting Ancient Greece.
We are in the midst of studying. More posts soon. You should study too, my disciples.
PEACE.
We feel bad about leaving this post so empty, so please enjoy this picture of a cow:
Tuesday, September 21, 2010
Unit Conversions In Ancient Greece
Here are your measurements:
Length = 20 arm spans
Width = 10 arm spans
If in one arm span, there are 10 thumbs, and the area of the field is 200 arm span squared, what would the area be in thumb squared?


